Windgap Medical

This Boston startup is working to develop an epinephrine auto-injector that’s 40 percent smaller than current auto-injectors, simple to use, and has a long shelf life. This new auto-injector stores the drug in a temp-stable, dry form until it’s needed. Upon activation, the device rapidly and automatically mixes the dry drug to form a solution when the cap is removed. According to Windgap’s CEO, the improved temperature stability means that if their epinephrine auto-injector is left in a hot car on a sunny day, it won’t have to be thrown away. Windgap’s product will be submitted for FDA review in 2018.
Details: windgapmedical.com
Epi-Pill

Researchers in Florida are working to develop a dissolvable pill that, when placed under the tongue, will reportedly deliver epinephrine as effectively as an auto-injector. While this product may be more portable, affordable and easier to use than an injector, it has yet to be tested on patients experiencing anaphylactic shock. Currently, they are still awaiting FDA approval to begin testing and trials.
Details: popsci.com
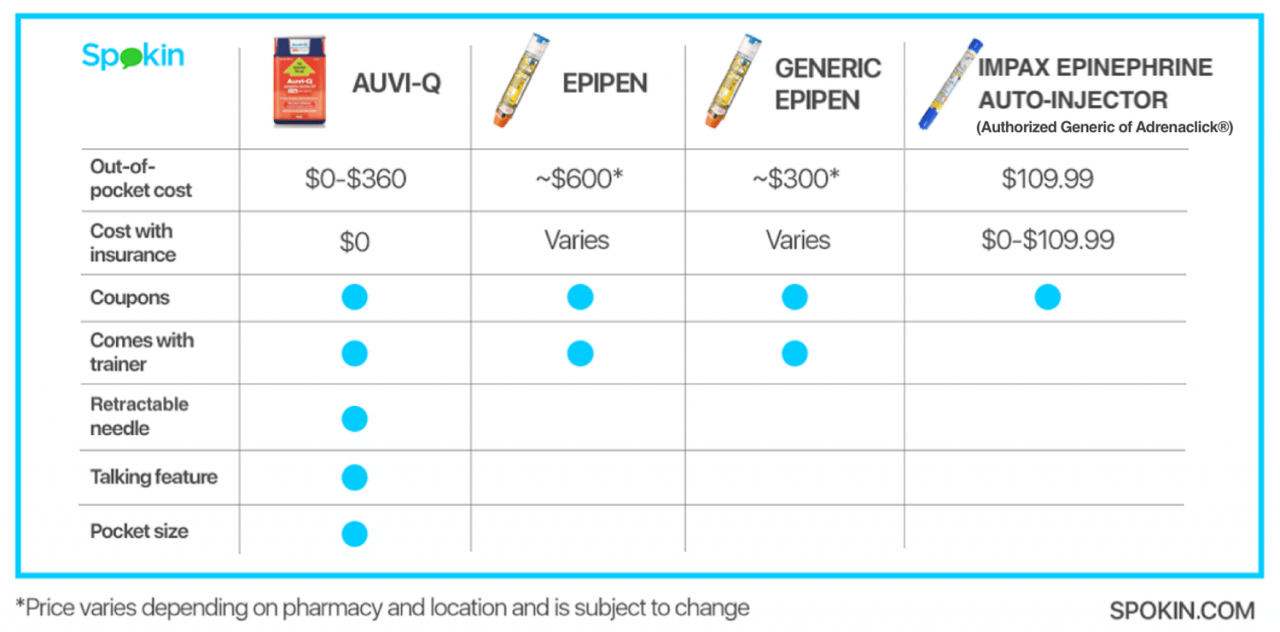
AUVI-Q

Since the 2015 recall of AUVI-Q, Kaleo took back the license to manufacture the popular auto-injector and invested in new systems to ensure “accurate, reliable and consistent delivery” from the product. In February 2017, AUVI-Q returned to the market with an unprecedented affordability plan. AUVI-Q is the same portable size and still features the unique voice prompt system that offers users step-by-step instructions. For more information, check out Spokin’s AUVI-FAQ and learn How To Get AUVI-Q For $O.
Details: auvi-q.com